Gay-Lussac's Law
Gay-Lussac's Pressure-Temperature Law is more often called Amontons' Law of Pressure-Temperature, because it was previously discovered by Guillaume Amontons between 1700 and 1702. The law states that pressure of a gas is directly related (proportional) to it's temperature. In other words, as the temperature goes up, the pressure also goes up. Increased temperature is the result of adding heat. This increases the kinetic energy of the gas particles, so they are moving faster. They strike each other and the walls of their container more often and with more force, so the pressure goes up.
Gay-Lussac's Law can be expressed in three ways:
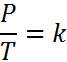 |
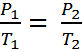 |
|
| Equation #1 | Equation #2 | Equation #3 |
P is the pressure of the gas
T is the absolute temperature of the gas (measured in kelvin).
k is a constant (which depends on the units of P & T)
If the ratio (or fraction) of P and T is always a constant, then we can use an initial pressure and temperature, P1 and T1 to solve for pressure or temperature at another set of conditions, P2 or T2. This is because if P1/T1 = k, it must also be true that,
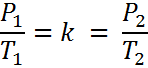
We can remove “k” and we get Equation #2. A little rearranging give us Equation #3.
It is important to note that you cannot solve this equation with temperatures in °C. You must convert to absolute temperature, or Kelvin (K). This is easy enough to do because, K = °C + 273. Also, pressures can be measured in any units, but whatever they are, both pressures must be in the same units.
Most students find the form of Equation #2 easiest to use. Consider the following word problem:
A gas with a pressure of 2.5 atmospheres and a temperature of 350 Kelvin is cooled to a temperature of 280 Kelvin. What is the new pressure?
First we identify the variables, P1=2.5 atm, T1=350K, T2 = 280 K and P2 is unknown. The temperatures absolutely must be in Kelvin. If the temperatures are in °C, simply add 273° to get the temperature in Kelvin. Now, we substitute into Equation #2 and get:
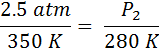
From here, we just cross-multiply the two circled numbers and then divide by the third
P2 = (280K x 2.5 atm) / 350K = 2.0 atm. You can subsitute and cross multiply to solve for any of the four variables.